
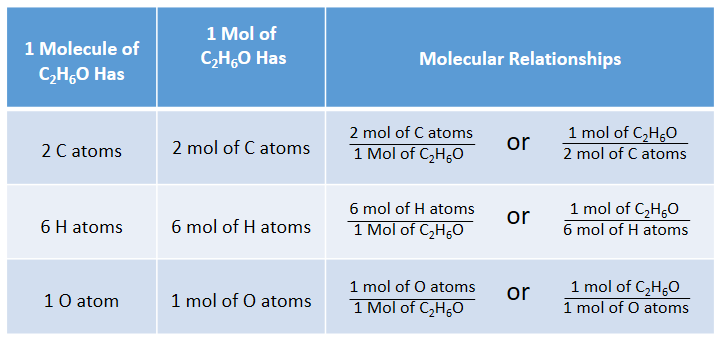
SOLVED: Calculate the number of molecules present in each of the following samples. a. 6.37 mol of carbon monoxide b. 6.37 g of carbon monoxide c. 2.62 × 10^-6 of water d.

Analysis of Two Definitions of the Mole That Are in Simultaneous Use, and Their Surprising Consequences | Journal of Chemical Education

Amazon.com: 6 in 1 Wooden Montessori Toys for 1 Year Old Whack a Mole Game Hammering Pounding Toy with Xylophone Carrot Harvest Game Learning Developmental Toys Toddler Activities Gift Ages 1 2 3 4 : Toys & Games

Gretchen Mol speaks with The Bare Magazine in NYC | The Notorious Bettie Page | Rounders — The Bare Magazine


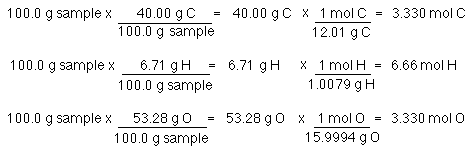


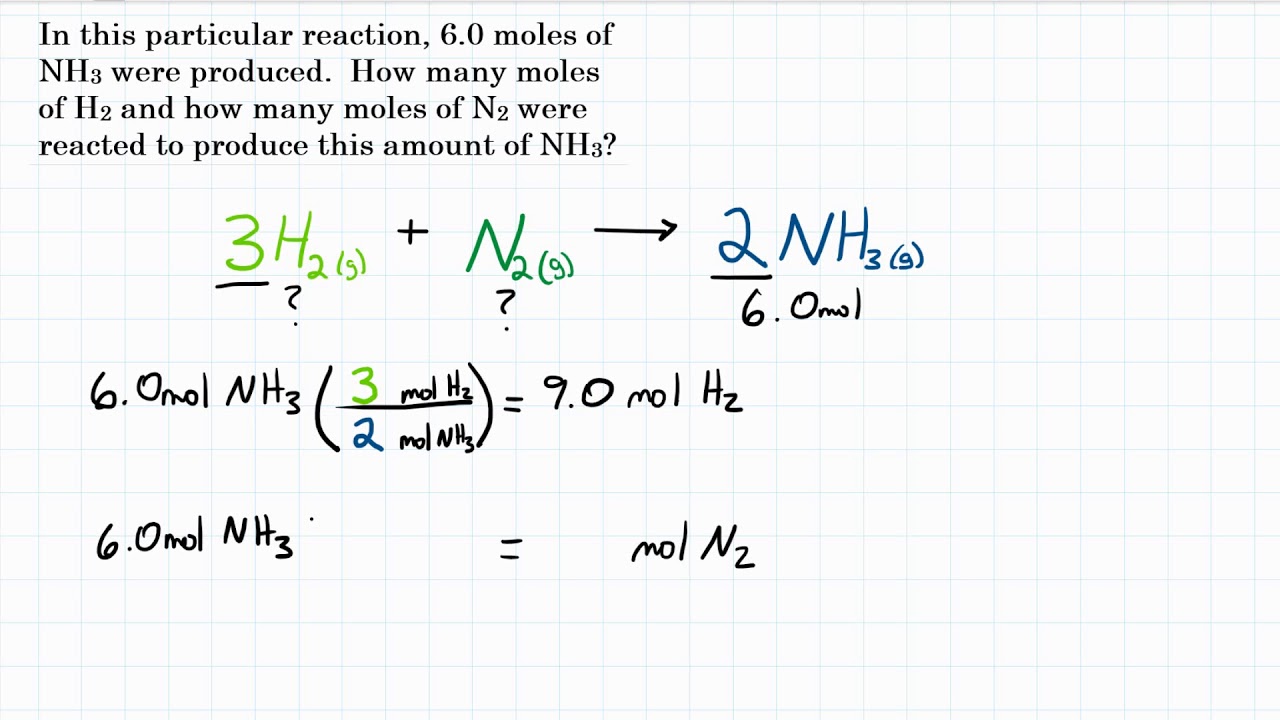




![Molecular orbitals diagrams of [Ti(H2O)6]3+ Molecular orbitals diagrams of [Ti(H2O)6]3+](https://image.slidesharecdn.com/molecularorbitalsdiagramsoftih2o631-211120080947/75/molecular-orbitals-diagrams-of-tih2o63-2-2048.jpg?cb=1666619489)
